
Lalwani SK, Agarkhedkar S, Sundaram B, Mahantashetti NS, Malshe N, Agarkhedkar S, Van Der Meeren O, Mehta S, Karkada N, Han HH, Mesaros N. Acute disease and/or fever at the time of enrolment.Administration of immunoglobulins and/or any blood products since birth or planned administration during the study period.Major congenital defects or serious chronic illness.History of any reaction or hypersensitivity likely to be exacerbated by any component of the vaccine.Family history of congenital or hereditary immunodeficiency.Any confirmed or suspected immunosuppressive or immunodeficient condition, based on medical history and physical examination.Evidence of previous diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and Haemophilus influenzae type b (Hib) vaccination or disease, with the exception of a birth dose of hepatitis B vaccine and oral poliovirus vaccine (OPV) as per local standard of care.Concurrently participating in another clinical study, at any time during the study period, in which the subject has been or will be exposed to an investigational or a non-investigational product.Administration of a vaccine not foreseen by the study protocol, within 30 days prior to the first study visit, or planned administration during the study period, with the exception of oral human rotavirus (HRV) vaccination which is allowed at any time during the study.Chronic administration of immunosuppressants or other immune-modifying drugs within six months prior to the first vaccine dose.Use of any investigational or non-registered product other than the study vaccines within 30 days preceding the first dose, or planned use during the study period.If ANYexclusion criterion applies, the subject must not be included in the study: The following criteria should be checked at the time of study entry. Born after a gestation period of at least 36 weeks.Healthy subjects as established by medical history and clinical examination before entering into the study.Written informed consent obtained from the parent(s)/LAR(s) of the subject. 
Subjects who the investigator believes that their parent(s)/legally acceptable representative(s) can and will comply with the requirements of the protocol.Documented administration of a hepatitis B vaccine dose at birth.A male or female between, and including, 6 and 10 weeks of age at the time of the first vaccination.Why Should I Register and Submit Results?Īll subjects must satisfy ALL the following criteria at study entry:.Any claims and inquiries should be addressed to the respective Merchant. Health.ESDlife is irresponsible to any loss, injury or law action caused by using this service/product. ESD Services Limited (“Health.ESDlife”) is not the service provider of this Service/Product. The Merchant is the service provider of this Service/Product.When there is any sign of symptom/disease in your health, please consult Doctor immediately for diagnosis and treatment. All health check/health screening services are not for the purpose of medical diagnostic or therapeutic purposes.In case of any disputes, the decision of Hong Kong Professional Vaccine Limited shall be final.Should there be any questions, please feel free to contact our consultants.Before service(s), customer should understand and agree with the service(s) details.All Health Check Services and/or Vaccination Services are not intended for medical diagnostic or therapeutic purposes.No refund is allowed once the Order has been confirmed.(Customers who purchased from Mawill have their validity extended to 6 months.) After which, the confirmation letter will be deemed invalid. 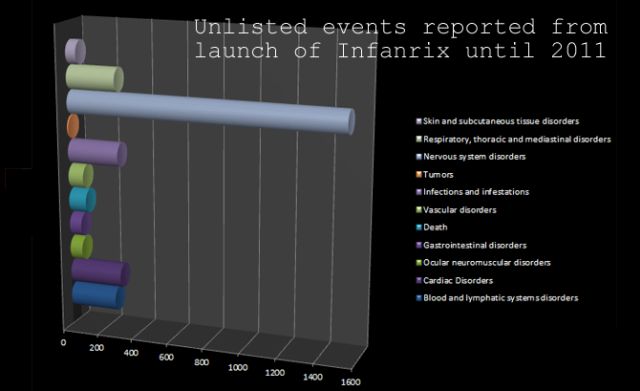
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
